This site is intended for
non-US residents only.
Please confirm your location.
Gelclair® for the management of painful symptoms of oral mucositis
Gelclair® is a viscous gel that used as a mouthwash, forms a protective film by adhering to the mucosa of the oropharyngeal cavity helping to provide pain relief in mouth lesions caused by:

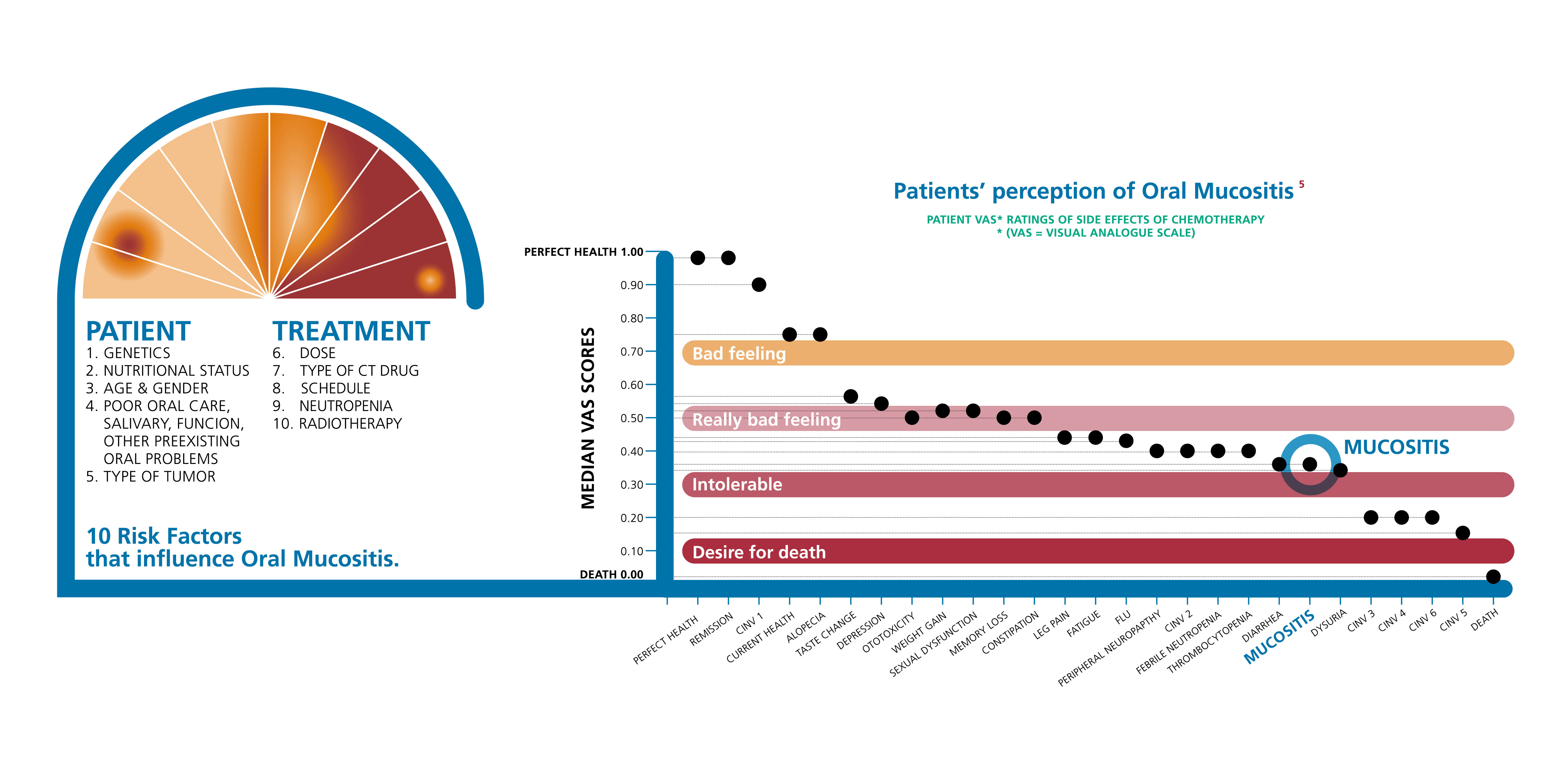
What is Oral Mucositis?
Oral mucositis is the acute inflammation and ulceration of the surface of the mouth and throat. Oral mucositis is a significant problem in patients receiving chemotherapy or radiotherapy. It is a painful condition that can limit nutritional intake and may be the cause for reducing, deferring, or stopping chemotherapy or radiotherapy and/or extend hospitalization1.
What is the effect of Oral Mucositis on patients’ daily life?
Patients may feel pain and troubles in:
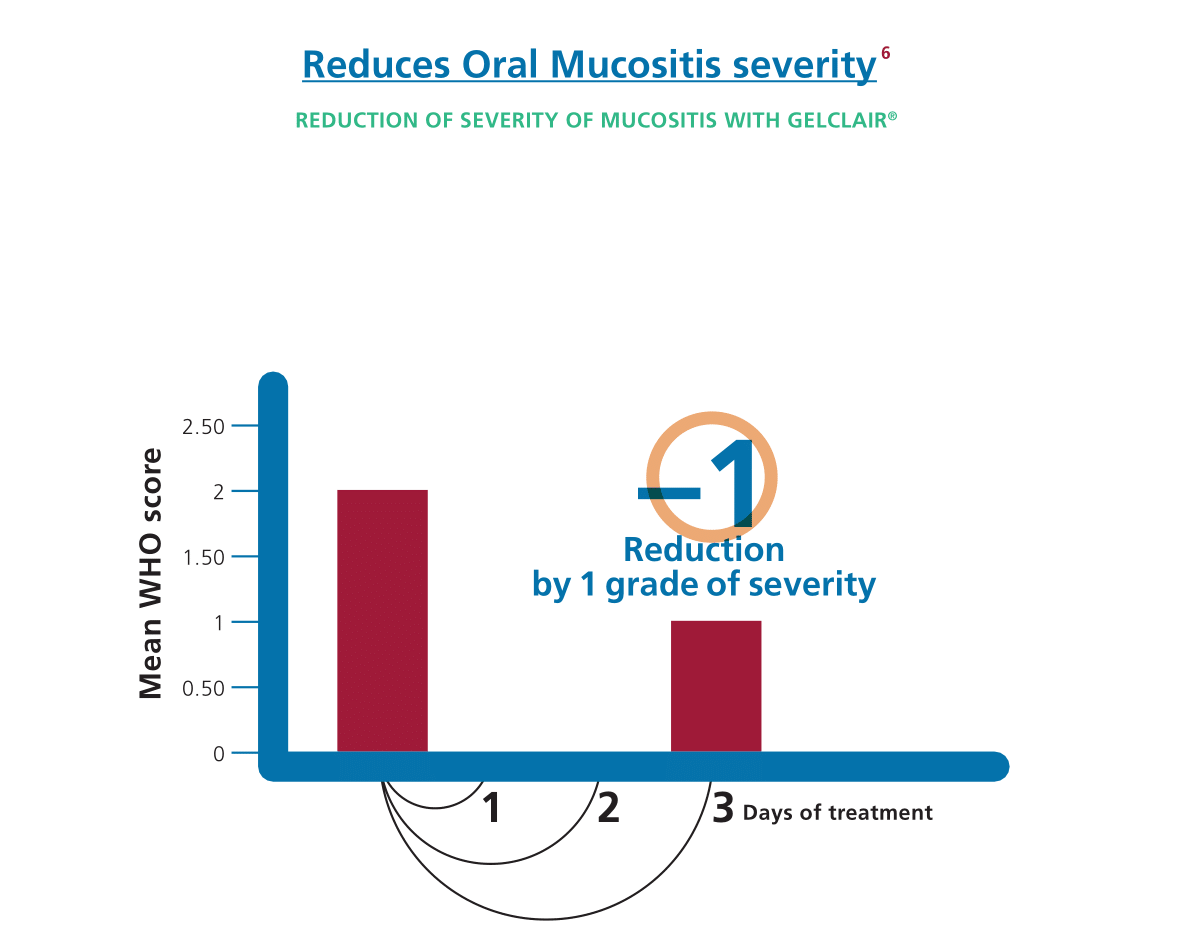
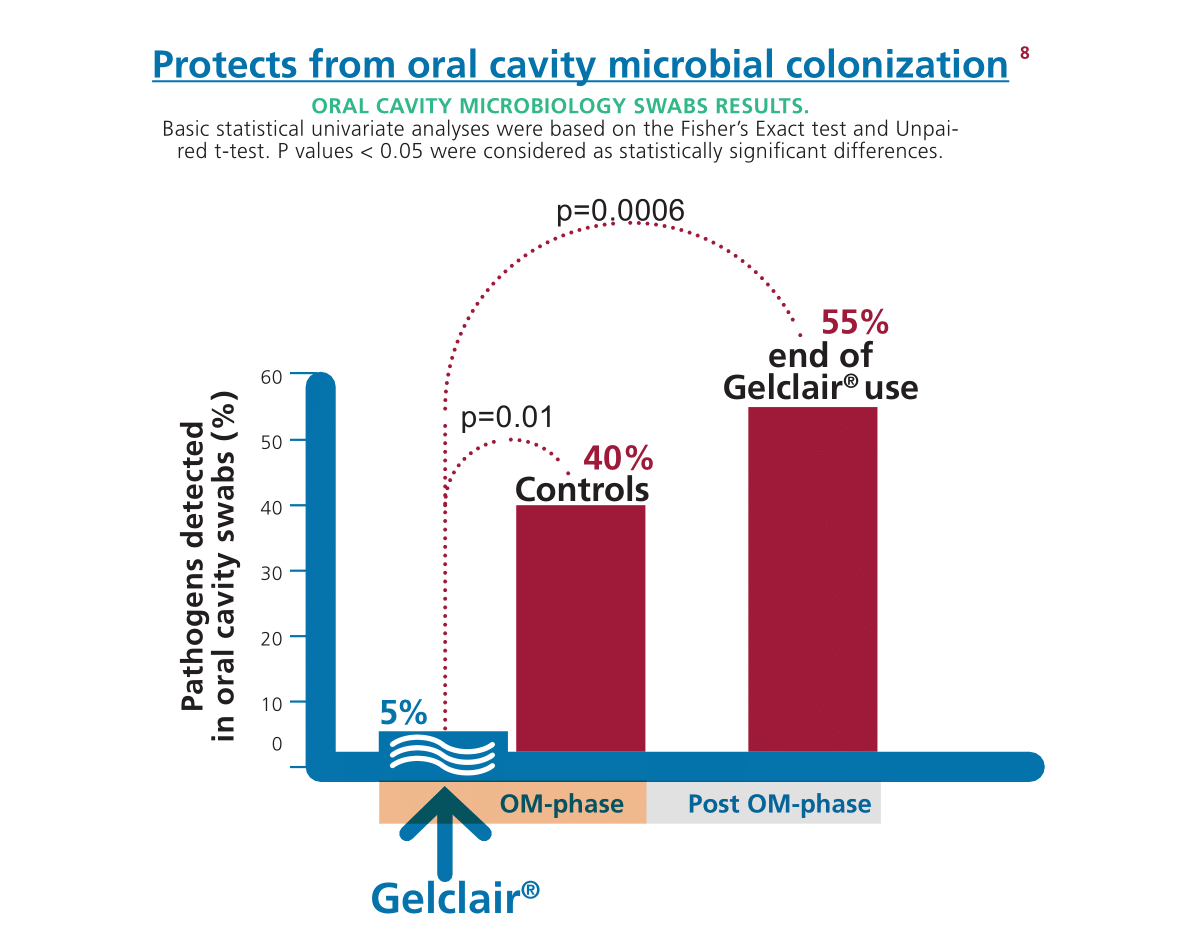
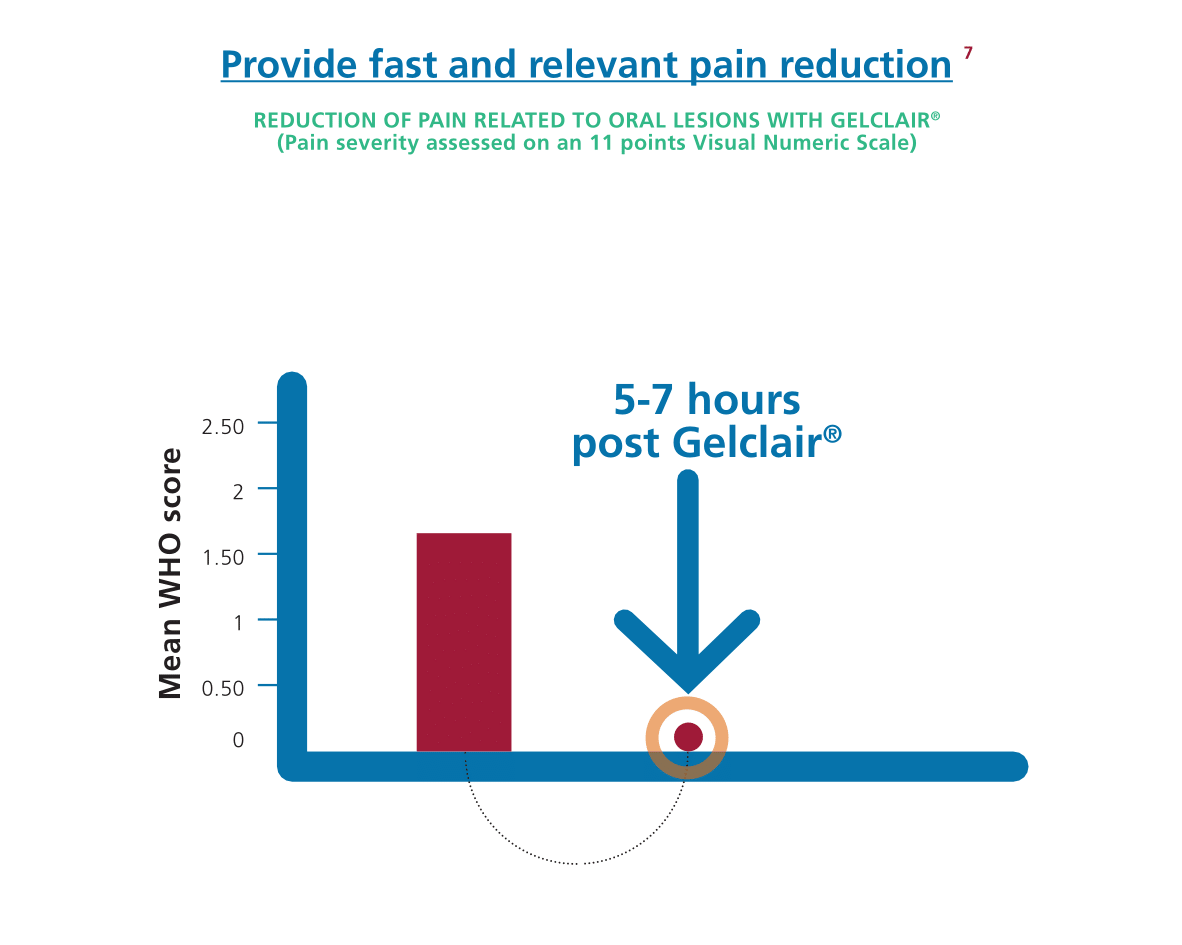
How does Gelclair® work?
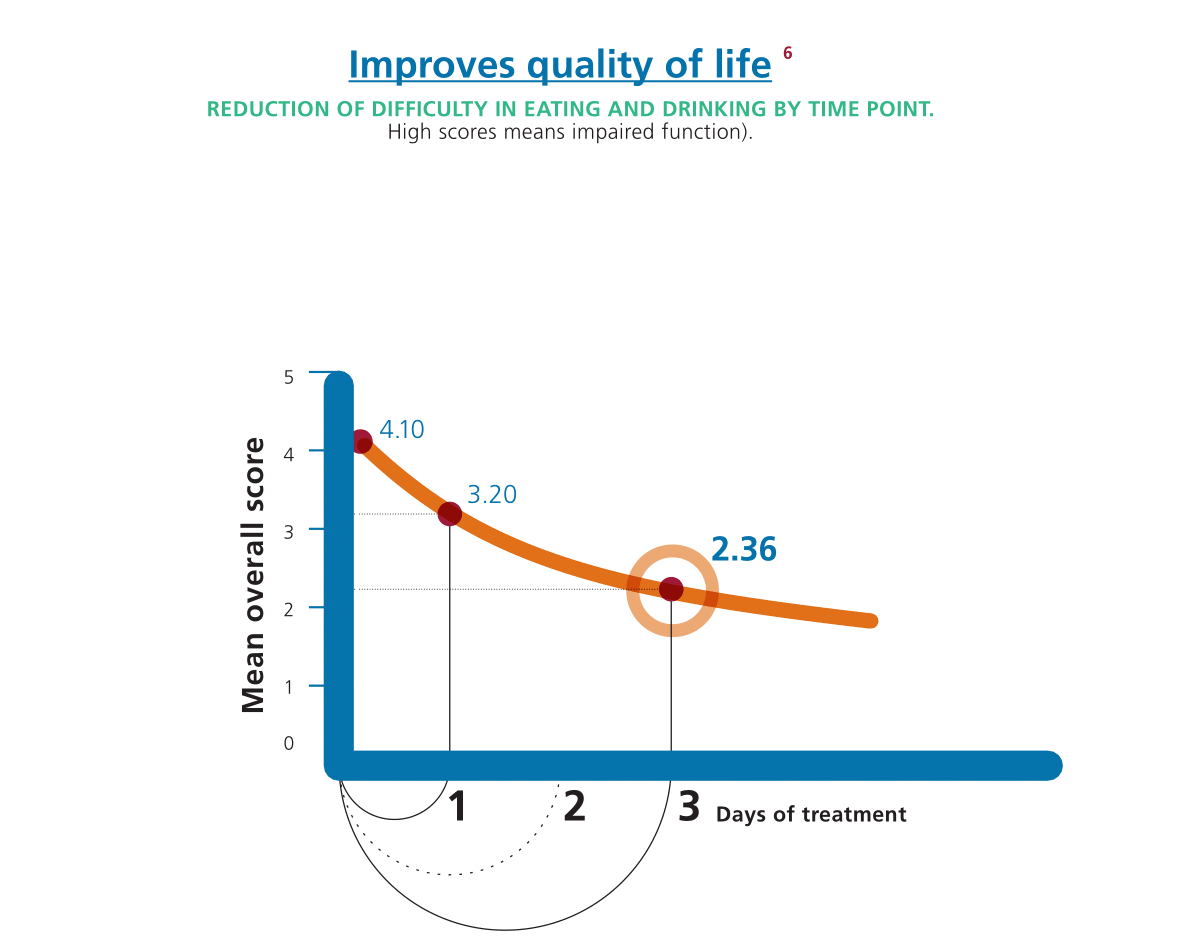
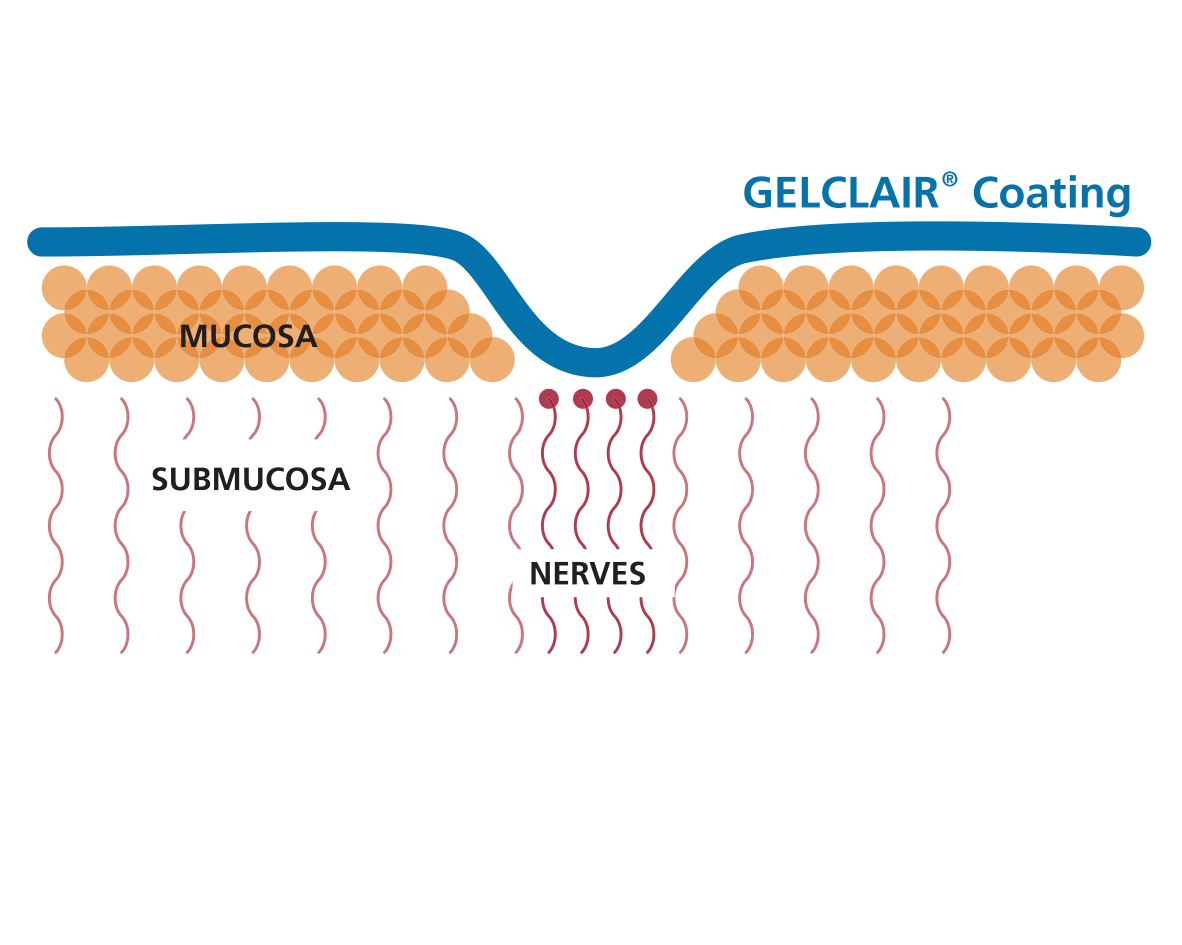
Gelclair® is a Poly-Vinyl-Pyrrolidone (PVP) and Hyaluronic Acid-based viscous oral gel that forms a protective film that adheres to the oral mucosa and is very well tolerated. The protective film shields the exposed nerves, due to oral lesions, for 5-7 hours from the first administration, offering a 92% of pain reduction2,3. Gelclair® can help patients maintain a normal dietary and fluid intake and undertake a full dose of chemotherapy and radiotherapy.
Gelclair® is dedicated to patient’s quality of life.
Patients all around the world use Gelclair® to manage Oral Mucositis because:
Have additional questions about Gelclair®? For more information, visit the FAQ page or click here to access the patient information leaflet

While treatment-related nausea, vomiting and cytopenia are the most common adverse events in oncology, mucositis has emerged as the most significant according to an NCCN task force10. OM can negatively affect diet, nutrition, oral hygiene, and quality of life11. Its adverse physical, social and psychological impacts on patients are manifest and include the need for total parenteral nutrition, higher risk of systemic infections, increased use of antifungals and opioid analgesics, increased hospitalisations with longer hospital stays, social isolation and depression due to the inability to talk and eat, subsequent dose reductions, treatment interruptions and discontinuations, and as a corollary to dose reductions and treatment interruptions/discontinuations, diminished anti-tumor responses and shorter survival10.
The economic consequences are far from trivial as well with the incremental cost exceeding $17,000 (USD) for patients with head and neck cancer12. Accordingly, drugs for the treatment of oral mucositis are required to minimise its clinical aggressiveness and improve the nutritional status, hydration, and quality of life of the affected patients13.